Your industries and services news from Hawaii
Provided by AGPCytokinetics Announces New Data Related to MYQORZO® (aficamten) at ESC Heart Failure 2026
| Multinational Real-world Data Reinforce Favorable Clinical Profile and Long-Term Safety |
SOUTH SAN FRANCISCO, Calif., May 11, 2026 (GLOBE NEWSWIRE) -- Cytokinetics, Incorporated (Nasdaq: CYTK) today announced the presentation of new data reinforcing the clinical profile of MYQORZO® (aficamten) at the European Society of Cardiology (ESC) Heart Failure 2026 Congress. The presentations include new analyses from SEQUOIA-HCM, the pivotal Phase 3 clinical trial of aficamten in patients with oHCM; MAPLE-HCM, the Phase 3 clinical trial of aficamten compared to metoprolol in patients with symptomatic obstructive HCM (oHCM); and FOREST-HCM, the open-label extension trial of aficamten.
Collectively, the new evidence expands understanding of the effectiveness of cardiac myosin inhibition with aficamten compared directly to metoprolol, a beta blocker, as well as specific safety and durability characteristics of MYQORZO across patient demographics, clinical, and economic subgroups.
“The breadth of research being presented at ESC Heart Failure 2026 reflects our commitment to deepening the scientific understanding of oHCM and heart failure,” said Stephen Heitner, M.D., Senior Vice President, Clinical Research and Development, Cytokinetics. “These new insights underscore the strength and consistency of the clinical profile of MYQORZO, adding to the growing body of real-world evidence informing physician treatment decisions.”
New Analyses Show MYQORZO Outperforms Metoprolol Across Sex and Doses
A dose-dependent analysis from MAPLE-HCM compared aficamten to the beta-blocker metoprolol in patients with symptomatic oHCM. Key findings showed significant improvements in exercise capacity, outflow gradients, and N-terminal pro-B-type natriuretic peptide (NT-proBNP) across all treatment doses. Conversely, metoprolol failed to show improvement in these outcomes regardless of the dose administered (Figure 1).
Figure 1
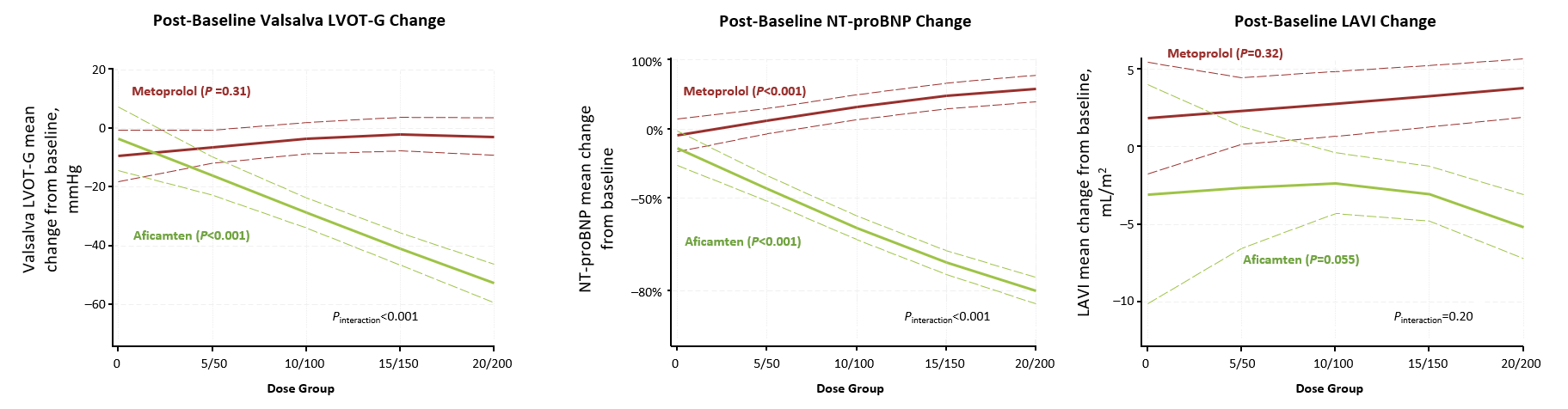
A secondary analysis of sex differences in MAPLE-HCM showed consistent benefits of MYQORZO in women. Despite women entering the trial (42%) with more severe baseline characteristics, MYQORZO delivered nearly identical improvements in peak oxygen consumption (pVO2) for both sexes (+2.2 mL/kg/min). Both groups also saw significant gains in Kansas City Cardiomyopathy Questionnaire Clinical Summary Score (KCCQ-CSS) and reductions in cardiac biomarkers and NT-proBNP.
Long-term Data Reinforce Safety and Efficacy Profile of MYQORZO
A prospective analysis of 122 patients who had interpretable ambulatory electrocardiogram (ECG) data at screening in the FOREST-HCM showed that long-term treatment with MYQORZO—up to 96 weeks—did not increase the incidence of arrhythmias in patients, including those who underwent withdrawal of beta-blocker therapy.
Among 122 patients, the incidence of non-sustained ventricular tachycardia (NSVT) on ambulatory ECG remained stable through 96 weeks compared with baseline, with no increase in atrial fibrillation (AF) episodes and no newly identified subclinical AF. There was also no increase in arrhythmia frequency among a subgroup of 16 patients who discontinued beta-blocker therapy during aficamten treatment. These results represent the first prospective analysis of ambulatory ECG monitoring in patients treated with a cardiac myosin inhibitor and are consistent with the low incidence of clinically detected arrhythmias previously reported for patients with oHCM treated with aficamten.
Additionally, an open-label extension study of aficamten in Chinese patients with symptomatic oHCM showed that aficamten was well tolerated using the same dosing strategy of individualized titration as was used globally. Through 48 weeks of treatment with aficamten, patients experienced no serious or severe treatment-emergent adverse events, no occurrences of LVEF <50% and no treatment discontinuations. Significant and durable improvements from baseline were observed in resting and Valsalva left ventricular outflow tract (LVOT) gradients (−40.3 mmHg and −45.6 mmHg, respectively; both p<0.001), New York Heart Association (NYHA) Functional Class ≥1 (15% p = 0.024), KCCQ-CSS (+8.3 points; p<0.001) and NT-proBNP (−59.2%; p<0.001).
New Evidence Indicates Positive Effects of MYQORZO on Left Atrium Remodeling and Atrial Mechanics
Expanded insights from SEQUOIA-HCM provide the first-ever analysis of the effect of aficamten on left atrial (LA) mechanics in oHCM. This analysis was performed to determine whether MYQORZO improved LA function in addition to previously demonstrated improvement in exercise capacity by relieving left ventricular outflow tract (LVOT). The new data showed that across the key clinical subgroups, MYQORZO, compared with placebo, significantly improved LA mechanics, which correlated with improvement in functional capacity in oHCM.
Among 269 patients, LA function was abnormal at baseline, with 94% of participants having reduced LA reservoir and conduit strain. Compared with placebo, aficamten significantly improved LA reservoir strain (treatment effect +2.9%; p=0.004) and absolute LA conduit strain (+2.2%; p=0.001) and reduced LA volume index (−3.5 mL/m2; p<0.001). These findings extend prior evidence that aficamten favorably remodels left atrial structure and suggest a beneficial effect on atrial mechanics.
Real-World Evidence Highlights Unmet Need and the Significant Burden of HCM
Multiple presentations provide new multinational real-world evidence and health economics and outcomes data underscoring the burden of HCM across disease subtypes and need for effective therapies that address the underlying disease mechanisms of HCM in adult and pediatric patients across the spectrum of symptomatic disease.
-
Work & Activity Impairment:
A survey of 273 patients in Italy, Spain and the United States revealed that patients with oHCM reported, on average, 26.6% activity impairment due to the disease. Full-time and part-time employed patients with HCM reported that 15.35% of their time at work was impaired due to HCM in the past 7 days and, on average, they missed 1.63% of work time. Activity impairment was significantly higher among those with oHCM compared with non-obstructive HCM (nHCM) (30.2% vs 19.2%; p=0.0003).
A separate analysis of 701 U.S. patients with HCM from the Adelphi Real World Disease Specific Programme™, showed that patients classified as NYHA II–IV Functional Class reported higher symptom burden, greater cardiovascular comorbidity, greater healthcare resource utilization (including emergency room (ER) visits, day visits and caregiver hours) and lower quality of life (EQ-5D-5L and EQ-VAS) compared with patients in NYHA Class I. Notably, patients in NYHA Class I continued to experience clinical symptoms and require medical care, underscoring the need for therapies that address the underlying mechanisms of HCM across the spectrum of symptomatic disease.
-
Limitations of Current Therapy:
An analysis of 723 patients with nHCM showed that despite 70% being on guideline-directed medical therapy such as beta-blockers, they continued to experience substantial clinical and economic burdens.
-
Clinical Burden of Pediatric Hypertrophic Cardiomyopathy:
A retrospective cohort study characterized the real-world epidemiology of pediatric HCM in the United States using administrative claims data from 6,093 children and adolescents diagnosed between 2016 and 2024. Approximately one in four children with HCM had obstructive disease (23.6%), and roughly one in four were symptomatic at presentation, underscoring the needs of the pediatric oHCM population that may be targeted by emerging disease-modifying therapies, including aficamten, which is currently under investigation in CEDAR-HCM, a Phase 3 trial in pediatric patients with oHCM.
A separate study analyzed the phenotypic distribution and clinical burden of pediatric HCM based on a longitudinal U.S. claims database, showing that pediatric patients with HCM experience substantial clinical burden. Obstructive HCM was associated with a higher cumulative risk of cardiovascular events compared with nHCM. These findings demonstrate clinically relevant heterogeneity in pediatric HCM and may inform risk mitigation strategies.
About MYQORZO® (aficamten)
MYQORZO® (aficamten) is a cardiac myosin inhibitor approved in the U.S., China and European Union for the treatment of symptomatic obstructive hypertrophic cardiomyopathy (oHCM). In patients with oHCM, myosin inhibition with MYQORZO reduces cardiac contractility and consequently, left ventricular outflow tract (LVOT) obstruction. MYQORZO was engineered to achieve a predictable exposure response, rapid onset of action and reversibility.1
Aficamten was studied in ACACIA-HCM, a completed Phase 3 clinical trial of aficamten in patients with nHCM, and is also under clinical investigation in CEDAR-HCM in a pediatric population with oHCM. Aficamten has not been deemed safe or effective for use in this patient population. In addition, aficamten is being studied in FOREST-HCM, an open-label extension clinical study. In addition, aficamten is being studied in FOREST-HCM, an open-label extension clinical study.
IMPORTANT SAFETY INFORMATION
| WARNING: RISK OF HEART FAILURE |
| MYQORZO reduces left ventricular ejection fraction (LVEF) and can cause heart failure due to systolic dysfunction. |
| Echocardiogram assessments are required prior to and during treatment with MYQORZO to monitor for systolic dysfunction. Initiation of MYQORZO in patients with LVEF <55% is not recommended. Decrease the dose of MYQORZO if LVEF is <50% and ≥40%. Interrupt the dose of MYQORZO if LVEF <40% or if the patient experiences heart failure symptoms or worsening clinical status due to systolic dysfunction. |
| Because of the risk of heart failure due to systolic dysfunction, MYQORZO is available only through a restricted program under a Risk Evaluation and Mitigation Strategy (REMS) called the MYQORZO REMS Program. |
CONTRAINDICATIONS
MYQORZO is contraindicated with concomitant use of rifampin.
WARNINGS AND PRECAUTIONS
Heart Failure
MYQORZO reduces cardiac contractility, which can reduce LVEF and cause heart failure.
Patients who experience a serious intercurrent illness (eg, serious infection) or arrhythmia (eg, new or uncontrolled atrial fibrillation) may be at greater risk of developing systolic dysfunction and heart failure.
Assess patients’ clinical status and LVEF prior to and during treatment and adjust the MYQORZO dose accordingly. New or worsening arrhythmia, dyspnea, chest pain, fatigue, leg edema, or elevations in N-terminal pro-B-type natriuretic peptide may be signs and symptoms of heart failure.
Initiation of MYQORZO in patients with LVEF <55% is not recommended.
MYQORZO REMS Program
MYQORZO is available only through a restricted program called the MYQORZO REMS Program, because of the risk of heart failure due to systolic dysfunction.
Notable requirements of the MYQORZO REMS Program include:
- Prescribers must be certified by enrolling in the MYQORZO REMS Program
- Patients must enroll in the MYQORZO REMS Program and comply with ongoing monitoring requirements
- Pharmacies must be certified by enrolling in the MYQORZO REMS Program and must only dispense to patients who are authorized to receive MYQORZO
- Wholesalers and distributors must only distribute to certified pharmacies
Further information is available at www.MYQORZOREMS.com, or at 1-844-285-7367.
Cytochrome P450 Interactions Leading to Heart Failure or Loss of Effectiveness
MYQORZO is metabolized primarily by CYP2C9, and to a lesser extent by CYP3A, CYP2D6, and CYP2C19 enzymes. Initiation of medications that inhibit multiple P450 pathways of MYQORZO elimination (eg, fluconazole, voriconazole, or fluvoxamine) or strong CYP2C9 inhibitors, and discontinuation of moderate-to-strong CYP3A inducers may lead to increased blood concentrations of aficamten and increase the risk of heart failure due to systolic dysfunction. Conversely, initiation of medications that induce P450 pathways of MYQORZO (eg, rifampin, moderate-to-strong CYP3A inducers) may lead to decreased blood concentrations of aficamten and potential loss of effectiveness. Assess LVEF 2 to 8 weeks after initiation of such inhibitors or after discontinuation of such inducers and adjust the dose of MYQORZO accordingly.
Advise patients of the potential for drug interactions. Advise patients to inform their healthcare provider of all concomitant medications prior to and during MYQORZO treatment.
ADVERSE REACTIONS
Hypertension (8% vs 2%) was the only adverse reaction occurring in >5% of patients and more commonly on MYQORZO than on placebo in the pivotal trial.
INDICATIONS AND USAGE
MYQORZO is indicated for the treatment of adults with symptomatic obstructive hypertrophic cardiomyopathy (oHCM) to improve functional capacity and symptoms.
Please see full prescribing information, including BOXED WARING, for MYQORZO.
About Hypertrophic Cardiomyopathy
Hypertrophic cardiomyopathy (HCM) is a disease in which the heart muscle becomes abnormally thick. HCM can be obstructive, when thickened muscle blocks blood flow, or non-obstructive, when blood flow is not blocked but heart function is still affected. In obstructive HCM, the thickening of cardiac muscle leads to the inside of the left ventricle becoming smaller, stiffer and less able to relax and fill with blood. Ultimately, HCM limits the heart’s pumping function, leading to reduced exercise capacity and a variety of symptoms.
HCM is the most common monogenic inherited cardiovascular disorder, with well over 300,000 patients diagnosed in the U.S.2 However, there are an estimated 400,000-800,000 additional patients who remain undiagnosed.3,4,5 Recent analysis of a large claims database indicates that approximately half of patients with HCM have obstructive HCM (oHCM) and half have non-obstructive HCM (nHCM).2
People with HCM are at high risk of also developing cardiovascular complications including atrial fibrillation, stroke and mitral valve disease.6 People with HCM are at risk for potentially fatal ventricular arrhythmias and it is one of the leading causes of sudden cardiac death in younger people or athletes.7 A subset of patients with HCM are at high risk of progressive disease leading to dilated cardiomyopathy and heart failure necessitating cardiac transplantation.
About Cytokinetics
Cytokinetics is a specialty cardiovascular biopharmaceutical company, building on its over 25 years of pioneering scientific innovations in muscle biology, and advancing a pipeline of potential new medicines for patients suffering from diseases of cardiac muscle dysfunction. Cytokinetics’ MYQORZO® (aficamten) is a cardiac myosin inhibitor approved in the U.S., Europe and China for the treatment of adults with symptomatic obstructive hypertrophic cardiomyopathy (oHCM). Following positive topline results in ACACIA-HCM, a Phase 3 clinical trial of aficamten in patients with non-obstructive HCM, the company is preparing to present the full results at an upcoming medical meeting and discuss them with the U.S. FDA and other regulatory authorities. Cytokinetics is also developing omecamtiv mecarbil, an investigational cardiac myosin activator for the potential treatment of patients with heart failure with severely reduced ejection fraction and ulacamten, an investigational cardiac myosin inhibitor for the potential treatment of heart failure with preserved ejection fraction, while continuing pre-clinical research and development in muscle biology.
For additional information about Cytokinetics, visit www.cytokinetics.com and follow us on X, LinkedIn, Facebook and YouTube.
Forward-Looking Statements
This press release contains forward-looking statements for purposes of the Private Securities Litigation Reform Act of 1995 (the “Act”). Cytokinetics disclaims any intent or obligation to update these forward-looking statements and claims the protection of the Act's safe harbor for forward-looking statements. Examples of such statements include, but are not limited to, statements, express or implied, related to Cytokinetics’ research and development activities; clinical trial initiation, design, enrollment, conduct, progress, continuation, completion, timing and results; regulatory submissions, review processes, approval timing and outcomes, including with respect to supplemental applications and approvals in jurisdictions outside the United States; the scope, expansion, modification, durability or continuation of labeling and promotional claims; commercial readiness, launch timing, market access and reimbursement; anticipated patient, prescriber and payer adoption; expectations regarding market opportunity, growth and market share; pipeline development and expansion into additional indications or geographies; access to and use of capital; and Cytokinetics’ business strategy, objectives and future plans. Such statements are based on management's current expectations and assumptions; however, actual results may differ materially due to various risks and uncertainties, including, but not limited to, uncertainties inherent in drug development and commercialization; the timing, conduct and outcomes of clinical trials; regulatory review and approval processes in the United States and other jurisdictions; differences in regulatory requirements, labeling, market access or promotional restrictions across jurisdictions; the ability to obtain, expand, maintain or continue desired labeling, promotional claims or commercial positioning for approved products; potential legal, intellectual property or regulatory constraints affecting commercialization and marketing claims; patient and prescriber acceptance of MYQORZO as compared to alternative therapies; the availability and terms of reimbursement from commercial and government payers; manufacturing, supply and distribution risks; competition; and the availability of sufficient capital to execute Cytokinetics’ business plans. These forward-looking statements speak only as of the date they are made, and Cytokinetics undertakes no obligation to subsequently update any such statement, except as required by law. For further information regarding these and other risks related to Cytokinetics’ business, investors should consult Cytokinetics’ filings with the Securities and Exchange Commission (the “SEC”).
CYTOKINETICS® and the CYTOKINETICS C-shaped logo are registered trademarks of Cytokinetics in the U.S. and certain other countries.
MYQORZO® is a registered trademark of Cytokinetics in the U.S. and the European Union.
Contact:
Cytokinetics
Diane Weiser
Senior Vice President, Corporate Affairs
(415) 290-7757
References
- Hartman JJ, Hwee DT, Roebrt-Paganin J, et al. Aficamten is a small-molecule cardiac myosin inhibitor designed to treat hypertrophic cardiomyopathy. Nat Cardiovasc Res. 2024;3(8) :1003-1016. doi:10.1038/s44161-024-00505-0
- Butzner M, et al. Epidemiology of Hypertrophic Cardiomyopathy in the United States From 2016 to 2023. JACC Adv. 2026. 2026;5(2):102552. doi:10.1016/j.jacadv.2025.102552
- CVrg: Heart Failure 2020-2029, p 44; Maron et al. 2013 doi:10.1016/S0140-6736(12)60397-3; Maron et al 2018 10.1056/NEJMra1710575
- Symphony Health 2016-2021 Patient Claims Data DoF;
- Maron MS, Hellawell JL, Lucove JC, Farzaneh-Far R, Olivotto I. Occurrence of Clinically Diagnosed Hypertrophic Cardiomyopathy in the United States. Am J Cardiol. 2016; 15;117(10):1651-1654.
- Gersh, B.J., Maron, B.J., Bonow, R.O., Dearani, J.A., Fifer, M.A., Link, M.S., et al. 2011 ACCF/AHA guidelines for the diagnosis and treatment of hypertrophic cardiomyopathy. A report of the American College of Cardiology Foundation/American Heart Association Task Force on practice guidelines. Journal of the American College of Cardiology and Circulation, 58, e212-260.
A photo accompanying this announcement is available at https://www.globenewswire.com/NewsRoom/AttachmentNg/5179347f-327c-44f4-9e9c-4e74b9ade881

Legal Disclaimer:
EIN Presswire provides this news content "as is" without warranty of any kind. We do not accept any responsibility or liability for the accuracy, content, images, videos, licenses, completeness, legality, or reliability of the information contained in this article. If you have any complaints or copyright issues related to this article, kindly contact the author above.


